| 217
LASIK Study |
As a LASIK trainer for over 6 years and one of the original pioneers of the Technolas 217 scanning laser, Dr. Machat was asked to be a lead international investigator for the Technolas laser FDA trials in January 1999. These trials are independently monitored and verified and are the most vigorously controlled clinical evaluations of an excimer laser. Patients must undergo 6 months of rigorous and controlled pre- and post-operative testing. While most clinical statistics quoted by surgeons are unsubstantiated, the FDA clinical outcomes are verified by independent monitors. There is no input from either the surgeon or the center.
The Technolas 217 scanning laser is equipped with PlanoScan 2000 software and utilizes a flying spot delivery system which scans a 2mm spot across an 8 to 9 mm zone to correct myopia, hyperopia and astigmatism. The laser is equipped with an active eyetracking system which follows the eye during treatment to improve clinical results and safety. It should be noted that the eyetracker was not utilized during FDA trials so patients now having the procedure with Dr. Machat routinely have even more advances available to them. |
|
| "The
LASIK clinical results demonstrated by Dr. Machat during
the trials were simply the best ever reported for LASIK
in such a study", stated Dr.
Stephen Slade, Medical Monitor for the FDA trial at
the European Society of Cataract and Refractive Surgery
Meeting in Vienna, Austria in September 1999. |
| CRS
LASIK premarket approval cohort data |
Summit |
Visx |
Dr.Machat |
| Eyes |
1013 |
723 |
213 |
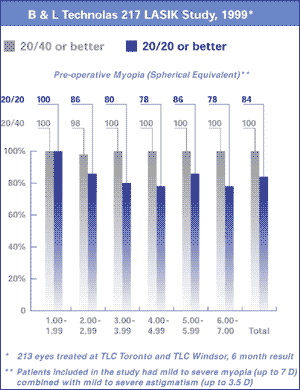
| Efficacy
- UCV 20/40 or better |
92% |
95% |
99.5% |
| UCVA
20/20 or better |
|
|
|
 7 D preop myopia
7 D preop myopia |
51% |
63% |
84% |
| Manifest
Refraction |
|
|
|
 0.5 D
0.5 D |
61% |
74% |
91.7% |
 1.0 D
1.0 D |
84% |
91% |
98.7% |
| Stability-<1-D
change from 3-6 months |
94% |
95% |
99.3% |
| Safety-BSCVA
lost 2 or more lines |
1.9% |
0.6% |
0% |
| Induced
Astigmatism |
1% |
0% |
0% |
| Adverse
Events |
<1% |
<1% |
0% |
|
 |
Clinical
results from other similar but less rigorous trials
are at left for comparison. (The CRS trials were multi-center
physician monitored LASIK trials which were submitted
to the FDA for LASIK approval of both the Summit and
Visx lasers.)
It is also important to note that during the clinical
study none of Dr. Machat's patients encountered any
corneal flap |
complications resulting in any loss of best corrected
vision. Therefore an exceptional safety record complimented
the superb clinical results.
Although the clinical study only evaluated mild to severe
myopia and astigmatism, Dr. Machat has been routinely
treating hyperopia, hyperopic astigmatism and mixed
astigmatism since 1996. |
| Dr.
Jeffery J. Machat Statistics |
| First
LASIK: 1994 |
| Total
number of LASIK procedures 1994-2000: 28,000 |
| Average
number of LASIK procedures over the past 3 years: 5,000/year
|
| Corneal
Flap Complications: < 1/2000 in the past two years |
|
|
|
|